Why Choose Cosette CDMO?
Cosette CDMO is a partner grounded in pharmaceutical practice and built for long-term reliability.
We offer proven systems, experienced people and a collaborative approach that keeps projects moving.
Embedded pharmaceutical expertise
Cosette CDMO launched in 2018 to make our internal expertise available to partners. We support semi-solids, liquids, suspensions and suppositories, applying the same quality systems that guide our own medicines.
Our aim is straightforward: dependable, compliant delivery from transfer through commercial supply.


Partnership that delivers
We take a hands-on approach. From the first transfer to commercial launch, dedicated project leads keep communication open and decisions quick.
Working with Cosette CDMO means your intellectual property (IP) stays protected, your product is prioritized and your timelines are respected. A dedicated project manager keeps progress visible at every stage and patient-first decision-making drives every milestone.
Ready to bring your product to life?
Proven capability across complex dosage forms
Cosette’s facilities support end-to-end processes for challenging formulations.
We manage formulation development, scale-up, packaging, stability testing and commercial release under one unified quality framework — ensuring smooth progression from R&D through commercial production.
Core dosage forms:
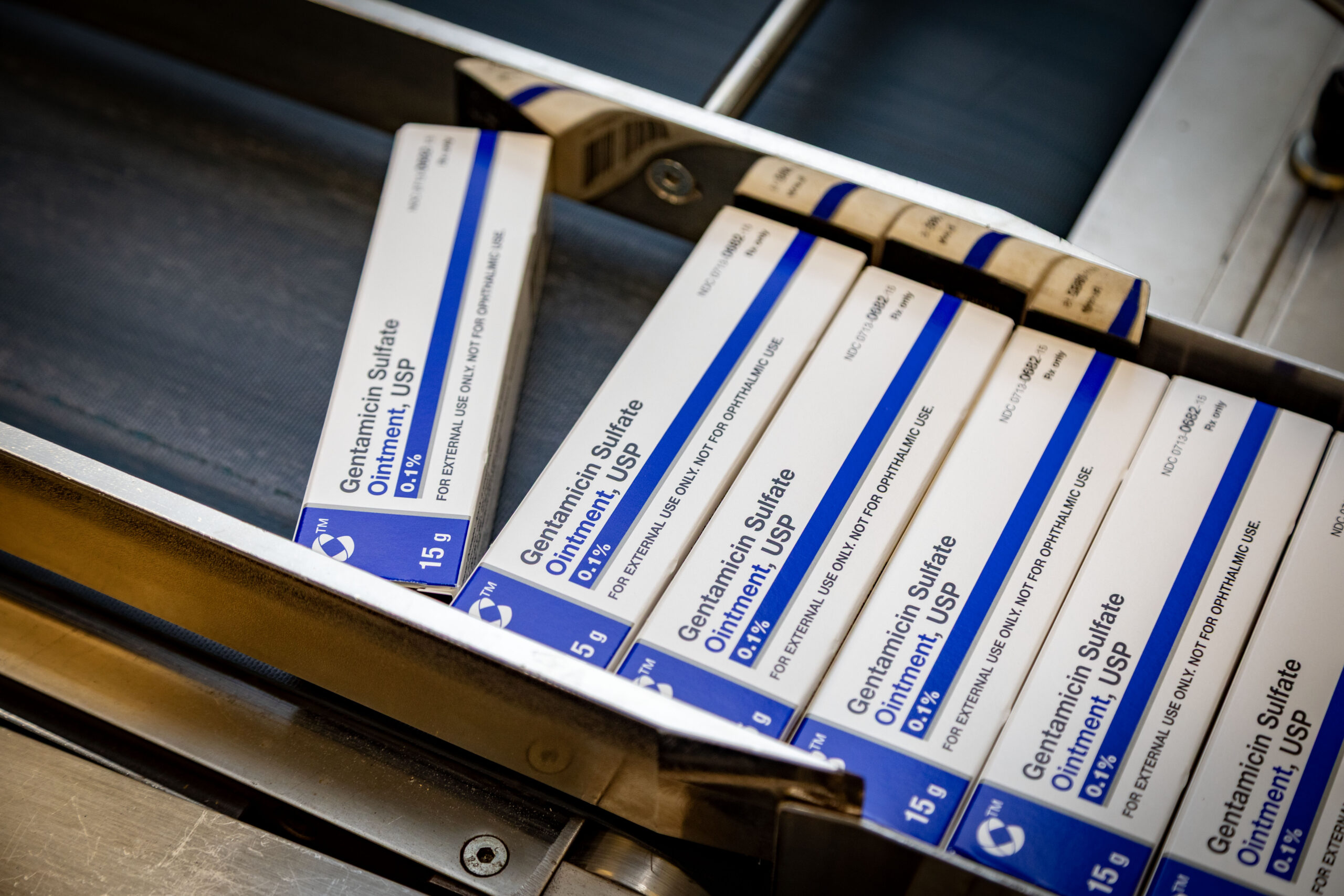
Semi-solids:
Creams, gels, ointments, lotions, shampoos

Liquids:
Oral solutions and suspensions
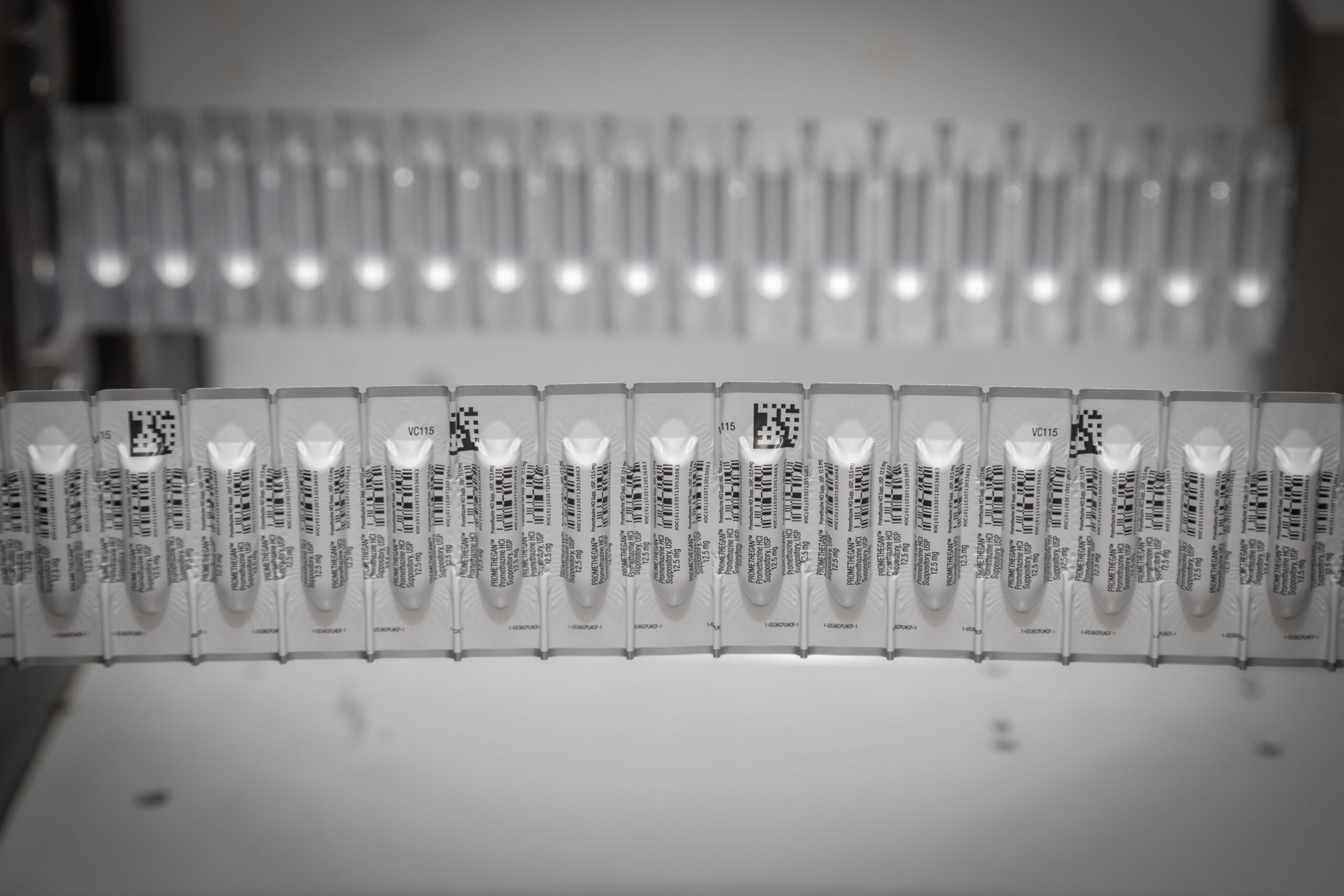
Suppositories:
One of the largest manufacturing operations in the U.S.
Quality and reliability at every stage
Our quality systems are embedded across development, technical services and manufacturing.
We operate to cGMP standards across every client program. Every process, investigation and validation follows the same standards that guide our own commercial products.

Corporate Headquarter and U.S. facility designed for efficiency and scale
Both sites operate to cGMP standards and maintain the infrastructure needed for reliable, traceable manufacturing.

Bridgewater, NJ – Corporate Headquarter
Serving as the central hub for our corporate leadership, quality, regulatory, sales, business development, and commercial operations. Our Bridgewater location anchors our commitment to innovation, operational excellence, and customer partnership across our CDMO network.

Lincolnton, NC - Manufacturing Hub
Spanning 12 acres and over 150,000 sq. ft., Lincolnton is the center of Cosette’s commercial operations. The facility brings together over 250 specialists who produce semi-solids, liquids, and suppositories at commercial scale, with integrated packaging, warehousing and controlled-storage operations that keep supply dependable and compliant.
Capabilities include
- Commercial scale dosage forms – creams, gels, ointments, lotions, shampoos, oral liquids (solutions and suspensions) and suppositories
- Technical capabilities include process optimization, technical transfer process support, product remediation, and reformulation
- Facility operates a full-service quality control laboratory, including chemistry and microbiology, as well as method development and validation capabilities
- Drug Enforcement Agency (DEA) – Schedule II through V (Raw materials and Finished goods)
- XP capabilities – Class I / Div. I
- Cold storage – 2 to 8 Celsius (Raw materials and Finished goods)
- End-to-end supply within Cosette’s U.S. network



