Please sign up to our news alerts. Once you’ve submitted your request, you will receive your activation link by email.
Cosette Pharmaceuticals Appoints Danielle DeBoer as Senior Vice President, Human Resources
Cosette Pharmaceuticals, Inc., a U.S.-based, branded specialty pharmaceutical company, announced today that it has appointed Danielle DeBoer as Senior Vice President, Human Resources.
Cosette Pharmaceuticals Expands Branded Portfolio through Acquisition of SYMPAZAN® and Additional Products from Assertio
Cosette Pharmaceuticals, Inc., a U.S.-based, branded specialty pharmaceutical company, announced today the closing of a definitive agreement to acquire the U.S. sales and distribution rights for a portfolio of seven branded products from Assertio Holdings, Inc. (Nasdaq: ASRT). The acquisition expands Cosette’s branded portfolio and adds the U.S. rights to SYMPAZAN®, a growing, patent-protected asset, further advancing Cosette’s disciplined growth strategy.
Cosette Pharmaceuticals Appoints Joseph Skelton as Senior Vice President, Corporate Development and Strategy
Cosette Pharmaceuticals, Inc., a U.S.-based, fully integrated, pharmaceutical company, announced today that it has appointed Joseph (Joe) Skelton as Senior Vice President, Corporate Development and Strategy.
Cosette Pharmaceuticals Announces the Approval and Launch of First Generic Version of CIPRO® HC (ciprofloxacin 0.2% HCl and hydrocortisone 1% otic suspension), with 180 days Competitive Generic Therapy (CGT) exclusivity
Cosette Pharmaceuticals, Inc., a U.S.-based, fully integrated pharmaceutical company, announced today that the U.S. Food and Drug Administration (FDA) has approved Cosette’s Abbreviated New Drug Application (ANDA) for the first generic version of CIPRO® HC (ciprofloxacin 0.2% HCl and hydrocortisone 1% otic suspension), with 180 days of Competitive Generic Therapy (CGT) exclusivity. Cosette has commenced commercial shipments, triggering the 180 days of market exclusivity.
Termination of Proposed Acquisition of Mayne Pharma
Cosette Pharmaceuticals, Inc. (Cosette), a U.S.-based, fully integrated pharmaceutical company, confirms that on 9 December 2025 it served a notice on Mayne Pharma Group Limited (Mayne Pharma, ASX: Ticker MYX) terminating the agreement between Cosette and Mayne Pharma under which Cosette was to acquire all the outstanding shares of Mayne Pharma (Scheme Implementation Deed). Cosette also notes the announcements from Mayne Pharma on 10 December 2025 and 11 December 2025 and confirms that on 11 December 2025 Mayne Pharma served its own notice of termination of the Scheme Implementation Deed on Cosette.
Cosette Pharmaceuticals Appoints Jeff Neubig as Vice President, Quality and Regulatory Affairs
Cosette Pharmaceuticals, Inc., a U.S.-based, fully integrated, pharmaceutical company, announced today that it has appointed Jeff Neubig as Vice President, Quality and Regulatory Affairs.
Cosette Pharmaceuticals Appoints Vincent Colicchio as Senior Vice President of Operations
Cosette Pharmaceuticals, Inc., a leading specialty pharmaceuticals company, today announced the appointment of Vincent (Vin) Colicchio as Senior Vice President of Operations, effective February 3, 2025. In this role, Vin will lead Cosette’s manufacturing, global supply chain, and operational strategy, ensuring efficiency, and supply continuity as the company continues its strong trajectory of growth and transformation.
Cosette Pharmaceuticals Appoints Brad Leonard as Vice President, Commercial Operations
Cosette Pharmaceuticals, Inc., a leading specialty pharmaceuticals company, today announced the appointment of Brad Leonard as Vice President, Commercial Operations. In this role, Brad will spearhead Cosette’s generics business, including Generics Sales, Marketing/Pricing, Customer Service, and Demand Planning, as the company continues its expansion in these markets.
Avista Capital Partners and Hamilton Lane Announce Partnership in Cosette Pharmaceuticals
Avista Capital Partners (“Avista”), a leading New York-based private equity firm focused exclusively on healthcare, and Hamilton Lane, a leading global private markets investment management firm (Nasdaq: HLNE), today announced that funds managed by Hamilton Lane have acquired a significant equity interest in Cosette Pharmaceuticals (“Cosette”), a US-based specialty pharmaceutical company, from Avista and its co-investors.
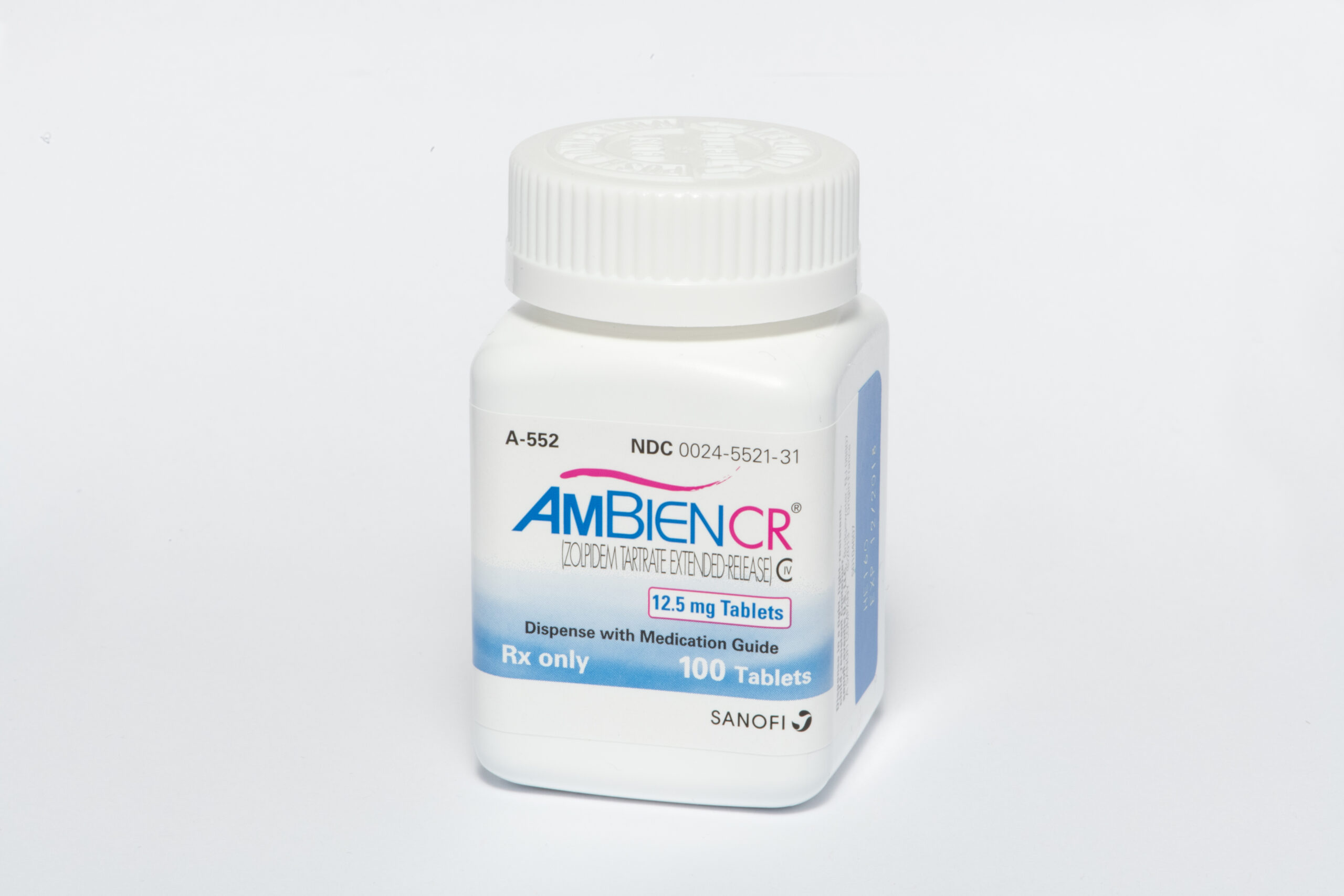
Cosette Pharmaceuticals Acquires Ambien® and Ambien CR® (Zolpidem Tartrate) Tabs from Sanofi US in the US Market
Cosette Pharmaceuticals, Inc. (“Cosette”), a US-based specialty pharmaceutical company, has completed the acquisition of Ambien® and Ambien CR® in the United States from Sanofi US.