Manufacturing capability across key dosage forms
Cosette specializes in dosage forms that demand technical skill and process stability.
Our facilities support commercial manufacturing of semi-solids, liquids, suspensions and suppositories, using equipment and controls designed for consistency at every scale.
Production spans pilot through full-scale campaigns, giving clients a single partner from transfer to commercial supply. Each batch follows defined processes for uniformity, yield and traceability.

Core capabilities:
With over 30 years of operational success and a proven regulatory track record, Cosette blends large-scale infrastructure with the responsiveness of a specialized CDMO.
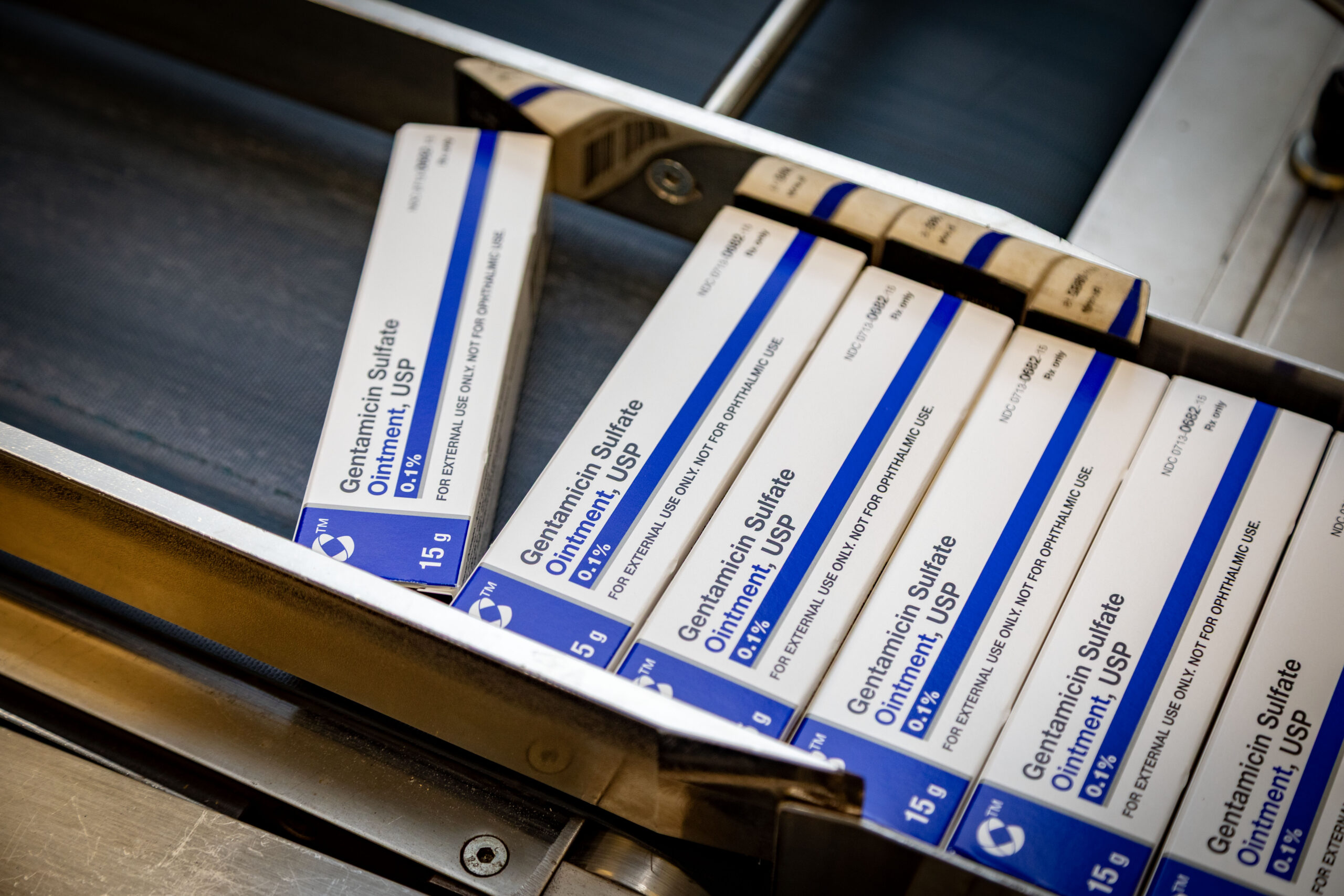
Semi-solids:
Creams, gels, ointments, lotions, shampoos

Liquids:
Oral solutions and suspensions
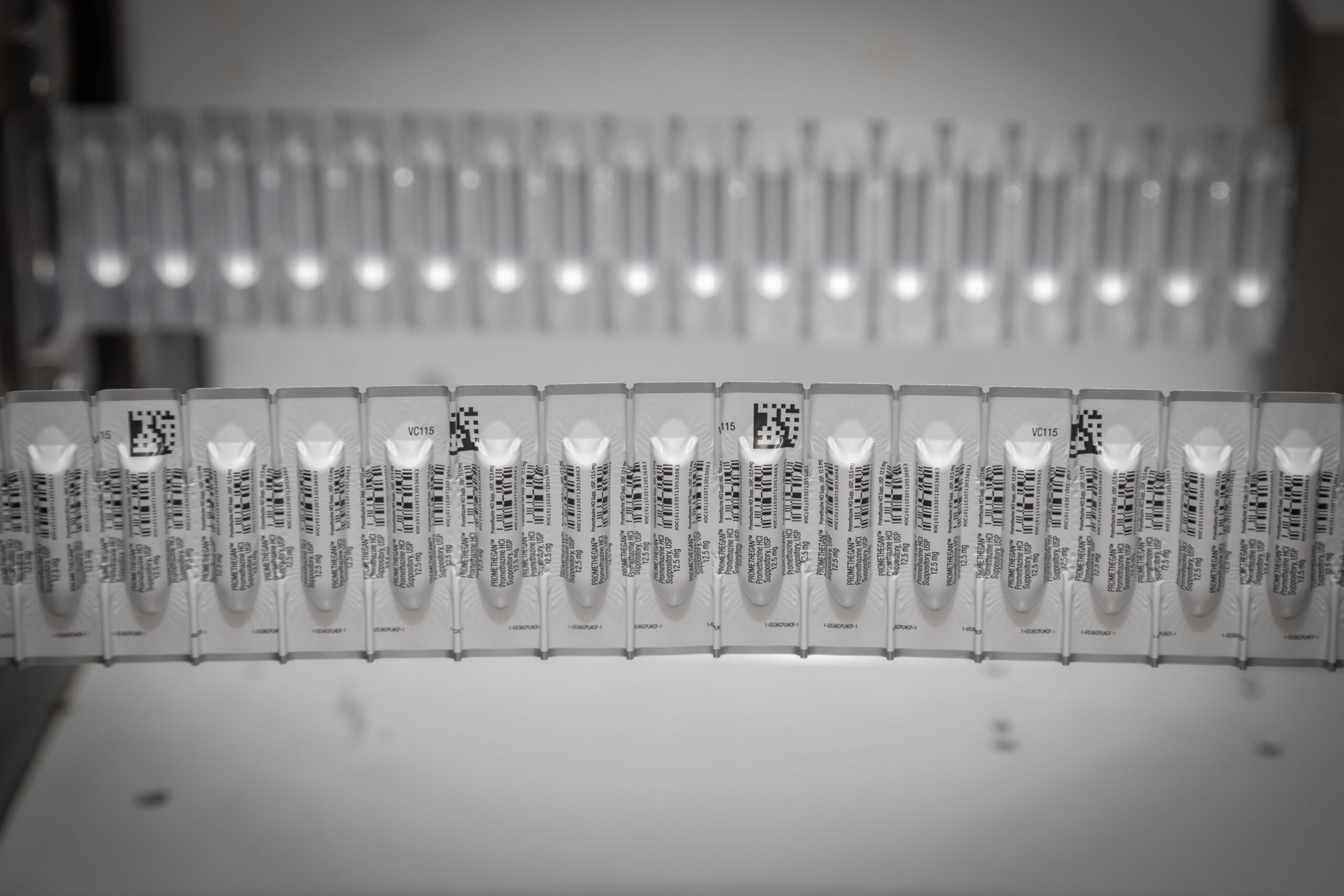
Suppositories:
One of the largest manufacturing operations in the U.S.
Discuss your manufacturing or packaging project.

Flexible operations that adapt to each product
Every product behaves differently during in manufacture, so each process is built to fit its physical and chemical profile.
Engineering and operations teams adjust parameters and validate changes to maintain throughput and reproducibility. The result is stable performance across runs and reliable timelines for release.
Projects benefit from continuous contact between Manufacturing, Technical Services and Quality, keeping reviews active and issues visible until every requirement is met.
“Our team has the expertise to modify, purchase, and qualify equipment to meet a client’s exact needs.”
— Mark Campbell, Director, EH&S, Engineering & Facilities Maintenance
Integrated packaging services
Packaging operations are part of the same quality system that governs manufacturing. Cosette handles bottles, tubes and suppository formats, supported by serialization and controlled storage for temperature-sensitive products.
Each configuration is validated for integrity, presentation and compliance before release. The packaging group works directly with Manufacturing and Quality to shorten change-control cycles and maintain full product traceability through distribution.
Additional strengths include:
- DEA Schedule II–V license for controlled substances
- In-house stability programs and environmental control
- Flexible labeling, cartoning, and shipping solutions

Corporate Headquarter and U.S. facility designed for efficiency and scale
Both sites operate to cGMP standards and maintain the infrastructure needed for reliable, traceable manufacturing.

Bridgewater, NJ – Corporate Headquarter
Serving as the central hub for our corporate leadership, quality, regulatory, sales, business development, and commercial operations. Our Bridgewater location anchors our commitment to innovation, operational excellence, and customer partnership across our CDMO network.

Lincolnton, NC - Manufacturing Hub
Spanning 12 acres and over 150,000 sq. ft., Lincolnton is the center of Cosette’s commercial operations. The facility brings together over 250 specialists who produce semi-solids, liquids, and suppositories at commercial scale, with integrated packaging, warehousing and controlled-storage operations that keep supply dependable and compliant.
Capabilities include
- Commercial scale dosage forms – creams, gels, ointments, lotions, shampoos, oral liquids (solutions and suspensions) and suppositories
- Technical capabilities include process optimization, technical transfer process support, product remediation, and reformulation
- Facility operates a full-service quality control laboratory, including chemistry and microbiology, as well as method development and validation capabilities
- Drug Enforcement Agency (DEA) – Schedule II through V (Raw materials and Finished goods)
- XP capabilities – Class I / Div. I
- Cold storage – 2 to 8 Celsius (Raw materials and Finished goods)
- End-to-end supply within Cosette’s U.S. network
A partner built for long-term delivery
Cosette offers the dependability of an established pharmaceutical organization and the responsiveness of a specialist CDMO.
Clients work with experienced teams who communicate clearly and maintain focus from transfer through release. Each project is managed with accountability and care to keep supply continuous and products secure.



